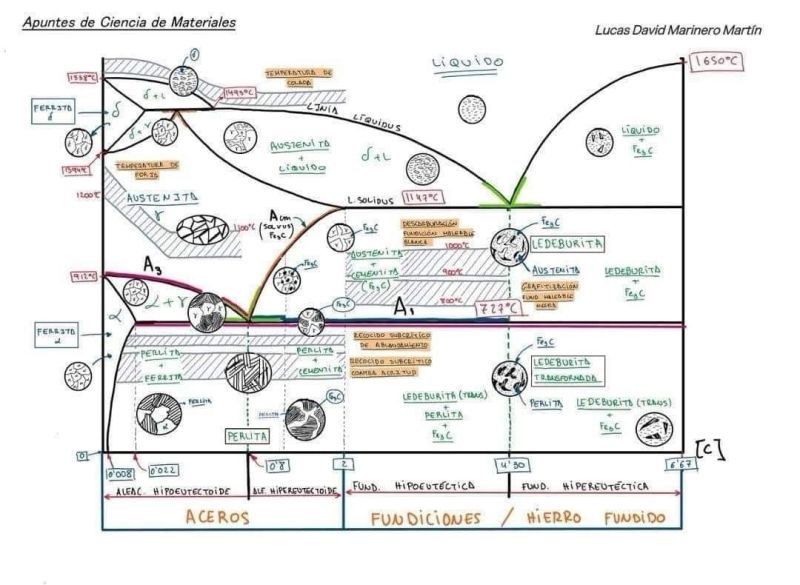
The Iron-Iron Carbide Phase Diagram is a fundamental tool in metallurgy, illustrating the phases and phase transformations of iron-carbon alloys as a function of temperature and carbon content. This diagram is critical for understanding the behavior of steels and cast irons during heat treatment, cooling, and alloy design. It maps the stability of various phases, including ferrite, austenite, cementite, pearlite, and ledeburite, under equilibrium conditions. This article provides a detailed explanation of the key phases and critical points in the diagram, offering insights into their properties and significance in materials science.
Key Phases in the Iron-Iron Carbide Phase Diagram
1. Ferrite (α-Fe)
Ferrite, also known as alpha iron, is a solid solution of carbon in body-centered cubic (BCC) iron. It is stable at temperatures below 912°C and has a very low carbon solubility, accommodating up to 0.022% carbon by weight at 727°C. Due to its BCC structure, ferrite is soft, ductile, and exhibits moderate strength, making it a key component in low-carbon steels. Its limited ability to dissolve carbon results from the small interstitial spaces in the BCC lattice, which restrict carbon atom incorporation.
2. Austenite (γ-Fe)
Austenite, or gamma iron, is a solid solution of carbon in face-centered cubic (FCC) iron. It exists in the temperature range of 727°C to 1493°C, depending on the carbon content of the alloy. Austenite has a significantly higher carbon solubility than ferrite, capable of dissolving up to 2.14% carbon at 1147°C. The FCC structure allows for greater interstitial space, enabling more carbon atoms to be accommodated. Austenite is stronger than ferrite but less magnetic, and it plays a critical role in heat treatment processes, as many phase transformations in steel occur from the austenite phase.
3. Cementite (Fe₃C)
Cementite, or iron carbide (Fe₃C), is a hard and brittle intermetallic compound with a fixed carbon content of 6.67% by weight. It forms in steels and cast irons when the carbon content exceeds the solubility limit of ferrite or austenite. Cementite is a key constituent in high-carbon steels and cast irons, contributing to their hardness but also increasing brittleness. Its presence in the microstructure significantly influences the mechanical properties of the material.
4. Pearlite
Pearlite is a lamellar microstructure composed of alternating layers of ferrite and cementite. It forms at the eutectoid point (0.76% carbon, 727°C) when austenite undergoes a transformation during cooling. The layered structure of pearlite results from the cooperative growth of ferrite and cementite, balancing the low-carbon ferrite with the high-carbon cementite. Pearlite is both strong and tough, making it a desirable constituent in many carbon steels used for structural applications.
5. Ledeburite
Ledeburite is a eutectic mixture found in iron-carbon alloys with carbon contents exceeding 2.14%, typically in cast irons. At the eutectic point (4.3% carbon, 1147°C), liquid transforms into a mixture of austenite and cementite. Below the eutectoid temperature, ledeburite may consist of cementite and a mixture of ferrite and austenite, depending on the temperature and cooling conditions. This phase is characteristic of high-carbon alloys and contributes to the unique properties of cast irons.
Critical Points in the Iron-Iron Carbide Phase Diagram
The Iron-Iron Carbide Phase Diagram contains several critical points where significant phase transformations occur. These points are essential for understanding the behavior of iron-carbon alloys during processing.
Eutectoid Point
The eutectoid point occurs at 0.76% carbon and 727°C. At this point, austenite transforms into pearlite, a mixture of ferrite and cementite, upon cooling. This transformation is critical in the production of carbon steels, as the formation of pearlite influences the mechanical properties such as strength and toughness. The eutectoid composition is often referred to as the “pearlite point” because it marks the carbon content at which pure pearlite forms.
Eutectic Point
The eutectic point is located at 4.3% carbon and 1147°C. At this point, the liquid phase transforms into a mixture of austenite and cementite, forming ledeburite. This reaction is characteristic of cast irons, which have higher carbon contents than steels. The eutectic transformation results in a microstructure that contributes to the hardness and wear resistance of cast irons, but it may also increase brittleness.
Peritectic Point
The peritectic point occurs at 0.16% carbon and 1493°C. At this point, a liquid phase reacts with delta ferrite (δ-Fe, a high- temperature BCC phase) to form austenite. The peritectic reaction is significant in low-carbon steels, as it marks the transition from the liquid and delta ferrite phases to the austenite phase during cooling. This transformation influences the microstructure and properties of the final material.
Significance of the Iron-Iron Carbide Phase Diagram
The Iron-Iron Carbide Phase Diagram is an indispensable tool for metallurgists and engineers. It provides critical information about the phases present in iron-carbon alloys under equilibrium conditions, guiding the design and processing of steels and cast irons. By understanding the stability and transformation of phases such as ferrite, austenite, cementite, pearlite, and ledeburite, engineers can tailor the mechanical properties of materials through controlled heat treatments and alloy compositions.
For example, low-carbon steels (less than 0.76% carbon) are typically soft and ductile due to their high ferrite content, making them suitable for forming applications. Medium-carbon steels, near the eutectoid composition, contain significant amounts of pearlite, offering a balance of strength and toughness for structural components. High-carbon steels and cast irons, with higher cementite and ledeburite content, are harder but more brittle, ideal for applications requiring wear resistance.
The Iron-Iron Carbide Phase Diagram is a cornerstone of materials science, providing a roadmap for understanding the phase transformations in iron-carbon alloys. The key phases—ferrite, austenite, cementite, pearlite, and ledeburite—each contribute distinct properties to steels and cast irons. The critical points, including the eutectoid, eutectic, and peritectic points, define the conditions under which these phases form and transform. By leveraging this diagram, engineers and metallurgists can optimize the processing and performance of iron-based materials for a wide range of industrial applications.